Human Cadherin-1 Protein
|
_Function: Cadherin-1 is a calcium dependent cell-cell adhesion
glycoprotein comprised of five extracellular cadherin
repeats, a transmembrane region and a highly conserved cytoplasmic
tail. Cadherins preferentially interact with themselves in a homophilic manner in connecting cells. Mutations in this gene are correlated with gastric, breast,
colorectal, thyroid and ovarian cancer. Loss of function is
thought to contribute to progression in cancer by
increasing proliferation, invasion, and/or metastasis [1]
|
Subcellular Location: Colocalizes with DLGAP5 at sites of cell-cell contact in intestinal epithelial cells. Anchored to actin microfilaments through association with alpha-, beta- and gamma-catenin. Sequential proteolysis induced by apoptosis or calcium influx, results in translocation from sites of cell-cell contact to the cytoplasm. Colocalizes with RAB11A endosomes during its transport from the Golgi apparatus to the plasma membrane. [3]
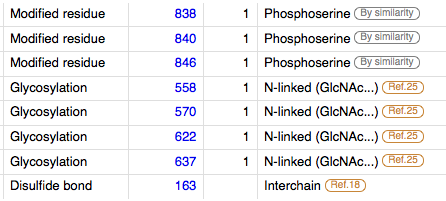
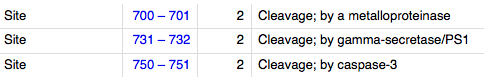
Post-translational Modifications: During apoptosis or with calcium influx, cleaved by a membrane-bound metalloproteinase, PS1/gamma-secretase and caspase-3 to produce fragments of about 38 kDa (E-CAD/CTF1), 33 kDa (E-CAD/CTF2) and 29 kDa (E-CAD/CTF3), respectively. Processing by the metalloproteinase, induced by calcium influx, causes disruption of cell-cell adhesion and the subsequent release of beta-catenin into the cytoplasm. The residual membrane-tethered cleavage product is rapidly degraded via an intracellular proteolytic pathway. Cleavage by caspase-3 releases the cytoplasmic tail resulting in disintegration of the actin microfilament system. The gamma-secretase-mediated cleavage promotes disassembly of adherens junctions. [cite] [cite] cite N-glycosylation at Asn-637 is essential for expression, folding and trafficking.[3]
Post-translational Modifications: During apoptosis or with calcium influx, cleaved by a membrane-bound metalloproteinase, PS1/gamma-secretase and caspase-3 to produce fragments of about 38 kDa (E-CAD/CTF1), 33 kDa (E-CAD/CTF2) and 29 kDa (E-CAD/CTF3), respectively. Processing by the metalloproteinase, induced by calcium influx, causes disruption of cell-cell adhesion and the subsequent release of beta-catenin into the cytoplasm. The residual membrane-tethered cleavage product is rapidly degraded via an intracellular proteolytic pathway. Cleavage by caspase-3 releases the cytoplasmic tail resulting in disintegration of the actin microfilament system. The gamma-secretase-mediated cleavage promotes disassembly of adherens junctions. [cite] [cite] cite N-glycosylation at Asn-637 is essential for expression, folding and trafficking.[3]
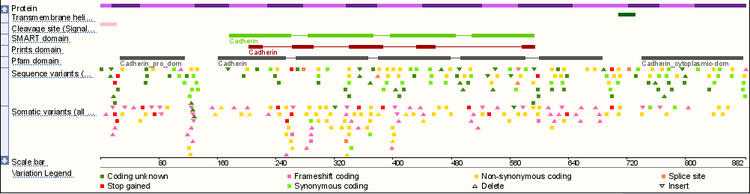
Human CDH1 Overview
Retrieved from useast.ensemble.org.